TEPMETKO demonstrated a manageable safety profile that maintained quality of life*1–4
Common AEs
Most common AEs were mild to moderate2
- Few discontinuations: only 14.7% of TrAEs led to discontinuation1
- TrAEs occurred in 91.7% of patients with a low rate of grade ≥3 (34.8%) and grade ≥4 (3.8%) TrAEs1
- TrAEs occurring in >20% of patients: peripheral edema (67.1%), hypoalbuminemia (23.6%), nausea (23.3%), diarrhea (22.4%) and increased blood creatinine (22.0%)1
- Favorable gastrointestinal tolerability: low rates of all-grade nausea (31%), diarrhea (29%) and vomiting (14%)5
- When required, AEs were effectively managed by simple dose modifications5
- 33.5% of patients experienced dose reduction and 43.1% treatment interruption1
TrAEs occurring in ≥10% of patients2
| SYSTEM ORGAN CLASS/AR | OVERALL (N=313), % | |
|---|---|---|
| ALL GRADES | GRADE ≥3 | |
| Metabolism and nutrition disorders | ||
| Hypoalbuminemia | 23.6 | 3.5 |
| Gastrointestinal disorders | ||
| Nausea | 23.3 | 0.6 |
| Diarrhea | 22.4 | 0.3 |
| Decreased appetite | 11.2 | 0.3 |
| Hepatobiliary, renal and urinary disorders | ||
| Increase in ALT | 14.1 | 2.2 |
| Increase in blood creatinine | 22.0 | 1.0 |
| General disorders | ||
| Peripheral edema | 67.1 | 11.2 |
Adapted from Mazieres et al. 20232
Quality of life
Quality of life was maintained throughout treatment3
Change in HRQoL scores from baseline**3
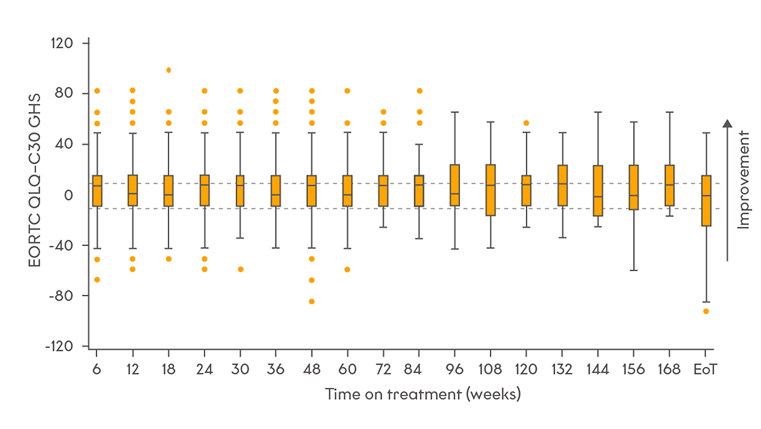
Adapted from Paik et al. 20233
Adapted from Paik et al. 20233
Dosing
Choose simplicity: TEPMETKO is the only MET inhibitor with once-daily oral dosing††5
Recommended dose modifications
If your patient requires an easy dose modification due to AEs, TEPMETKO is the only MET inhibitor that offers a prescription-free drop from two tablets to one5
Dose modification table
| ADVERSE REACTION | SEVERITY | DOSE MODIFICATION |
|---|---|---|
| Interstitial lung disease (ILD) | Any grade | Withhold TEPMETKO if ILD is suspected. Permanently discontinue TEPMETKO if ILD is confirmed |
| Increased ALT and/or AST without increased total bilirubin | Grade 3 | Withhold TEPMETKO until recovery to baseline ALT/AST. If recovered to baseline within 7 days, then resume TEPMETKO at the same dose; otherwise resume TEPMETKO at a reduced dose |
| Grade 4 | Permanently discontinue TEPMETKO | |
| Increased ALT and/or AST with increased total bilirubin in the absence of cholestasis or hemolysis | ALT and/or AST greater than 3 times ULN with total bilirubin greater than 2 times ULN | Permanently discontinue TEPMETKO |
| Increased total bilirubin without concurrent increased ALT and/or AST | Grade 3 | Withhold TEPMETKO until recovery to baseline bilirubin. If recovered to baseline within 7 days, then resume TEPMETKO at a reduced dose; otherwise permanently discontinue |
| Grade 4 | Permanently discontinue TEPMETKO | |
| Other adverse reactions | Grade 2 | Maintain dose level. If intolerable, consider withholding TEPMETKO until resolved, then resume TEPMETKO at a reduced dose |
| Grade 3 | Withhold TEPMETKO until resolved, then resume TEPMETKO at a reduced dose | |
| Grade 4 | Permanently discontinue TEPMETKO |
Managing peripheral edema
Peripheral edema is a class effect – identifying and managing it early on helps to maximize treatment benefit6,7
Amongst the 313 patients involved in the VISION study, peripheral edema was mostly mild to moderate; 11.2% of patients experienced a grade 3 event and none experienced a grade 4 event.1,6
Manage peripheral edema with a multimodal approach to help patients stay on TEPMETKO for as long as needed4,6,8
Effective management of peripheral edema includes:
- Prevention
- Intervention
Incorporating preventative measures at the start of therapy can help patients derive maximum benefit from TEPMETKO1,6
Help patients stay on TEPMETKO for as long as possible with individualized AE management strategies6,8,9
Tailored communication can empower patients to make informed choices, help mitigate the impact of AEs on their QoL and improve treatment adherence6